1
Biomicromolecules and biomacromolecules
1. Biomicromolecules are small-sized simple chemicals that have a low molecular weight, higher solubility and simple conformation.
2. They may be organic or inorganic and include minerals, sugars, amino acids and nucleotides.
3. Biomacromolecules are large-sized complex chemicals that have high molecular weight, low solubility and complex configuration.
They belong to carbohydrates, lipids, proteins and nucleic acid.
2
Nucleic acids
- Nucleic acids are biopolymers, or large biomolecules, essential for all known forms of life. Nucleic acids include DNA (deoxyribonucleic acid) and RNA (ribonucleic acid).
3
Organization of proteins
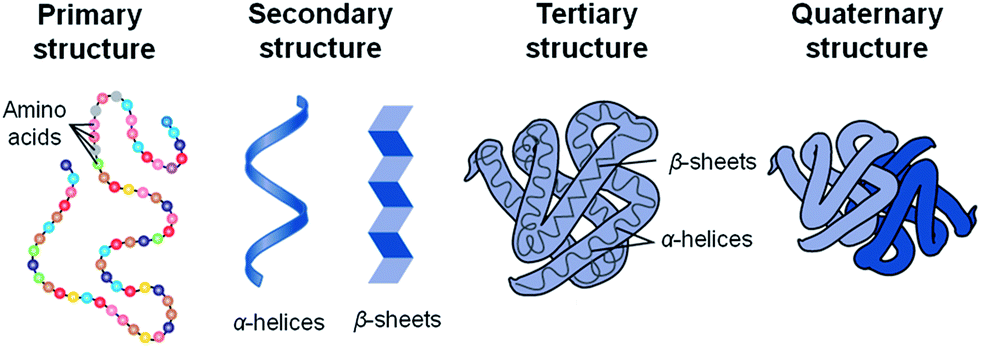
Proteins have four levels of organization: primary, secondary, tertiary, and quaternary.
1. The primary level of organization is simply the order of amino acids in the peptide chain. For example, Phe-Ala-Met-Leu-Gln-Trp-Glu-Ile.
2. Secondary structures are 2-dimensional structures formed due to hydrogen bonding between the hydrogen of amine groups and oxygen of the carbonyl groups. The two common secondary structures are the alpha-helix and the beta-sheet.
3. The tertiary structure involves the three-dimensional folding of a protein due to interactions of amino acid side chains. Where secondary structure was a result of hydrogen bonds between peptide groups, tertiary structure is a result of side chains interactions.
4. Quaternary structure involves the association of two or more polypeptides into functional proteins.
4
Peptide formation

1. Proteins are formed from amino acids, and the bond that connects them is called a peptide bond.
2. Peptide bonds are formed between the amine group of one amino acid and the carboxyl group of another.
5
Glycosidic bond
- A glycosidic bond or glycosidic linkage is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
6
Components of nucleotide

There are only three components to a nucleotide
1. A sugar (called deoxyribose)
2. A Phosphate (1 phosphorus atom joined to 4 oxygen atoms)
3. One of 4 bases (Adenine, Guanine, Cytosine, Thymine)
7
Nucleotide and nucleoside
1. A nucleoside consists of a nitrogenous base covalently attached to a sugar (ribose or deoxyribose) but without the phosphate group.
2. For example, cytidine, uridine, adenosine, guanosine, thymidine and inosine.
3. A nucleotide consists of a nitrogenous base, a sugar (ribose or deoxyribose) and one to three phosphate groups.
4.For example, 5′-uridine monophosphate.
8
Characteristics of an enzyme
Characteristics of an enzyme are as follows:
1. Destroys on heating.
2. Acts on one kind of substance.
3. Forms the same end product from the same substrate.
4. Speeds up the reaction.
5. Can be used again and again.
6. Acts best at a particular pH.
7. Acts best at a narrow temperature range.
9
Classification of enzymes
The main features of the classification proposed by the enzyme commission are as follows:
- All enzymes are divided into six major classes, viz. oxidoreductases, transferases, hydrolases, lyases, isomerases and ligases.
- Each major class of enzyme is divided into sub-classes and sub-sub-classes on the basis of the nature of the individual transformations involved.
- Each enzyme has a systematic code number (E.C).
10
Enzyme action by lowering activation energy

1. The graph above shows how the activation energy is lowered in the presence of an enzyme (blue line) that is doing the catalysis, exemplified by the carbon anhydrase reaction.
2.The transition state is usually the most unstable part of the reaction since it is the one with the highest free energy. The difference between the transition state and the reactants is the Gibbs free energy of activation, commonly known as activation energy.
3. Enzymes (blue line) change the formation of the transition state by lowering the energy and stabilizing the highly energetic unstable transition state. This allows the reaction rate to increase, but also the back reaction occurs more easily.